
Interest in retatrutide has grown quickly over the last two years, especially within metabolic and obesity research. What began as early discussion around next-generation GLP-related compounds has now turned into a broader scientific conversation about multi-pathway signaling, long-term metabolic outcomes, and what the future of peptide research may look like.
For researchers in Southern Utah, Washington County, and St. George, retatrutide often comes up alongside questions about regulatory status, published data, and why this compound has generated so much attention compared to earlier GLP-1 or dual-agonist research.
This article is intended to provide a research-focused, non-medical overview of retatrutide. It summarizes what has been published so far, explains why the compound is scientifically interesting, and outlines where the research appears to be heading — without making medical claims, recommendations, or human-use guidance.
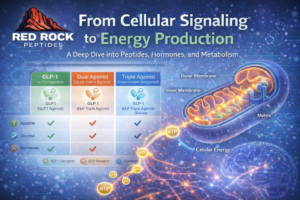
Retatrutide, also known by its research identifier LY3437943, is an investigational peptide being studied primarily for obesity and cardiometabolic outcomes. What makes retatrutide distinct is its design as a triple hormone receptor agonist, meaning it targets three metabolic signaling pathways at the same time:
Glucagon-Like Peptide-1 (GLP-1) receptors
Glucose-Dependent Insulinotropic Polypeptide (GIP) receptors
Glucagon receptors
Earlier generations of metabolic peptides typically focused on one pathway, most commonly GLP-1. More recent research expanded into dual-agonist compounds that activate both GLP-1 and GIP. Retatrutide represents the next step in that progression by adding glucagon receptor activity into the same molecule.
The scientific rationale behind this approach is that metabolism, appetite regulation, and energy balance are governed by multiple overlapping systems, not a single hormone signal. Triple-agonist research attempts to evaluate how coordinated receptor activation may influence overall metabolic outcomes.
Much of the attention around retatrutide comes from results published in a Phase 2 clinical trial in The New England Journal of Medicine.
That study, titled “Triple–Hormone-Receptor Agonist Retatrutide for Obesity,” evaluated adults with obesity and reported substantial average reductions in body weight across several study arms when compared with placebo.
You can read the full peer-reviewed publication here:
https://www.nejm.org/doi/full/10.1056/NEJMoa2301972
From a research standpoint, what mattered most was not just the magnitude of weight change, but the breadth of data generated, including:
Dose-response relationships
Tolerability patterns
Discontinuation rates
Metabolic and biomarker trends
These results do not establish long-term safety or clinical outcomes, but they provided a strong scientific rationale for expanded Phase 3 research programs.
GLP-1 research has already reshaped metabolic science by demonstrating how appetite signaling and glycemic control can be influenced pharmacologically. Dual-agonist research added another layer by examining how GLP-1 and GIP interact.
Triple-agonist research introduces a broader question: what happens when energy balance, appetite signaling, and metabolic regulation are all influenced simultaneously?
From a scientific perspective, this raises new areas of inquiry, including:
Energy expenditure and metabolic efficiency
Fat mass versus lean mass changes
Adaptive responses over longer study periods
Interactions between hormonal pathways
Retatrutide has become a focal point in this discussion because it is one of the most advanced triple-agonist compounds currently in large-scale human trials.
It is important to be clear about regulatory standing.
Retatrutide is not FDA approved and remains an investigational drug. Any legitimate human research involving retatrutide must occur within registered clinical trials under regulatory oversight.
The official regulatory authority in the United States is the U.S. Food and Drug Administration, and as of now, retatrutide has not received approval for any medical indication.
For researchers and readers looking for verified information about active studies, the most reliable source is ClinicalTrials.gov, which serves as the public registry for study design, endpoints, enrollment criteria, and trial status.
Examples of registered retatrutide studies include:
These listings provide transparent insight into what researchers are studying and what questions Phase 3 trials are designed to answer.
Phase 3 trials go beyond early efficacy signals. They are designed to answer questions that smaller or shorter studies cannot fully address, such as:
How durable weight changes are over longer periods
Cardiovascular and renal outcomes
Broader population safety data
Long-term tolerability and discontinuation trends
These trials are being conducted by Eli Lilly and Company, which has publicly outlined multi-year research programs focused on obesity and cardiometabolic disease.
The outcomes of these trials will largely determine whether retatrutide ever moves forward in the regulatory process.
Rather than framing retatrutide around individual use, responsible discussion focuses on research populations.
Clinical trials have evaluated or are evaluating retatrutide in populations such as:
Adults with obesity or overweight status
Individuals with metabolic risk factors
Cardiometabolic outcome cohorts
These groupings allow researchers to study statistical trends, biological mechanisms, and safety signals at the population level, which is fundamentally different from making recommendations for individuals.
As with any investigational compound, retatrutide research includes extensive safety monitoring. Trials track adverse events, laboratory values, tolerability, and participant withdrawal.
Because long-term data are still being collected, definitive safety conclusions cannot yet be drawn. This is a normal part of the research process and one of the reasons regulatory approval requires large, extended trials.
Caution, transparency, and continued data collection are essential parts of responsible peptide research.
One of the most important effects of retatrutide is how it has influenced future research directions.
Strong early trial signals often lead to:
Exploration of next-generation multi-agonist peptides
New delivery mechanisms and dosing strategies in research
Expanded investigation into metabolic maintenance and long-term outcomes
In this way, retatrutide represents not only a single investigational compound, but also a broader shift in how metabolic peptide research is being approached.
As interest in retatrutide has grown, informal discussions have emerged across online forums, social platforms, and community-driven websites where individuals speculate about dosing strategies and usage patterns.
Researchers are aware that these conversations exist, not because they serve as guidance, but because they highlight public curiosity and the gap between investigational research and approved medical use.
It is important to understand that these discussions:
Are not peer-reviewed
Are not standardized
Are not part of regulated clinical research
Do not replace clinical trial data
Media coverage has noted that investigational compounds like retatrutide are already being discussed outside of clinical settings, despite not being FDA approved. These discussions highlight the importance of clear regulatory boundaries and responsible research practices.
In simple terms, researchers are studying retatrutide because it appears to act on more than one metabolic pathway at the same time. Earlier GLP-1–based compounds mainly focus on appetite and blood sugar regulation, which can support weight loss but sometimes raise questions about fatigue, muscle loss, or metabolic slowdown. Retatrutide’s triple-pathway design has led researchers to explore whether it may influence weight reduction, exercise capacity, glucose regulation, and muscle retention differently than earlier GLP approaches. While no conclusions can be drawn outside of clinical trials, the research interest centers on whether targeting multiple signals at once could better support fat loss while preserving lean tissue and metabolic function, particularly in the context of obesity and type 2 diabetes research.
Southern Utah has quietly developed a growing base of independent researchers, academic collaborators, and laboratory professionals with interest in peptide and metabolic research. As investigational compounds like retatrutide move through larger clinical trial programs, curiosity around their mechanisms and future implications naturally extends beyond major research hubs.
For many researchers in St. George, Hurricane and surrounding Washington County areas, interest in retatrutide is not about immediate application, but about understanding where metabolic peptide research is headed next. Triple-agonist compounds represent a meaningful shift in how researchers think about appetite signaling, energy balance, and long-term health and metabolic outcomes. That shift has created demand for deeper study, comparison, and ongoing exploration.
Local researchers often face practical challenges when sourcing research materials, including long shipping timelines, inconsistent documentation, and unclear research-only positioning. As investigational peptides gain attention nationally, those challenges become more noticeable at the local level.
This is why local, research-focused peptide sourcing matters. Reliable access, clear documentation, and a strict research-only approach allow investigators to focus on study design and analysis rather than logistics.
As a Southern Utah–based research peptide supplier, we work directly with researchers who prioritize documentation, consistency, and scientific integrity. Our role is not to interpret outcomes or make claims, but to support ongoing research efforts with reliable, research-grade materials and transparent standards.
As research into retatrutide and next-generation metabolic peptides continues to evolve, the future of this field will be shaped not only by large clinical trials, but also by researchers like you who are exploring these compounds at a deeper, mechanistic level. Many investigators are interested in building on existing data, asking new questions, and contributing to the broader scientific conversation around multi-agonist peptides and metabolic regulation.
In Southern Utah and beyond, continued investigation, responsible sourcing, and a clear research-only approach allow researchers to take part in advancing understanding while remaining aligned with scientific and regulatory standards. Retatrutide represents one chapter in a much larger story, one that will continue to unfold as researchers explore where this class of compounds may lead next.