
Interest in semaglutide has expanded rapidly across the United States, including in St. George and surrounding Washington County communities such as Washington, Hurricane, Santa Clara, and Ivins.
From a scientific standpoint, semaglutide has drawn attention due to its role in GLP-1 receptor activity and its evaluation in multiple large-scale clinical trials.
This article explains:
• What semaglutide is
• What clinical studies have evaluated
• Why it has received national medical attention
• How it differs from other incretin-based compounds
• The distinction between prescription medications and research materials
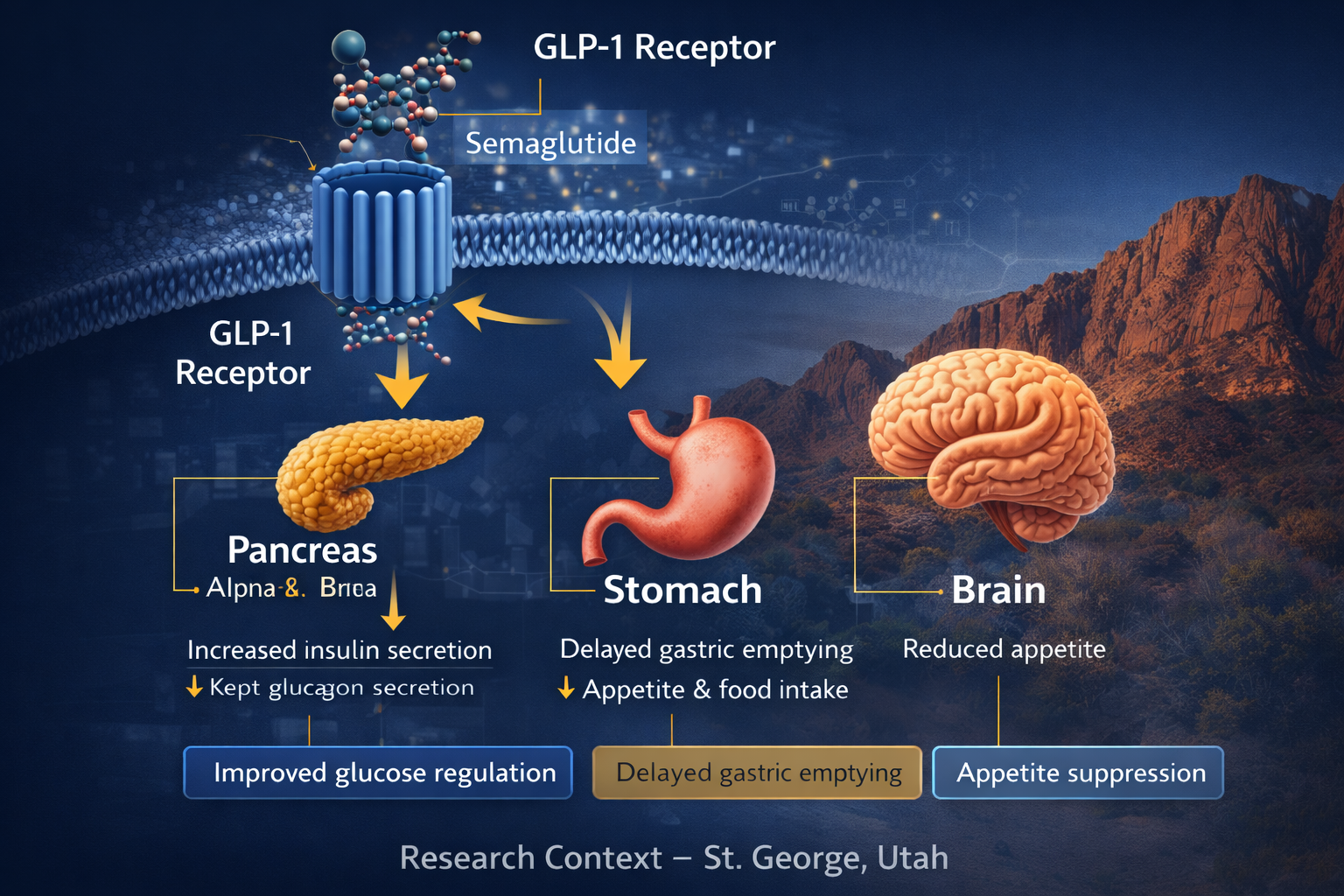
Semaglutide is a glucagon-like peptide-1 (GLP-1) receptor agonist.
GLP-1 is a hormone involved in:
• Glucose-dependent insulin signaling
• Gastric emptying regulation
• Appetite-related signaling pathways
• Metabolic feedback mechanisms
Pharmaceutical formulations of semaglutide have received FDA approval for specific medical indications under physician supervision.
Semaglutide gained widespread visibility after large randomized clinical trials evaluated it for:
• Type 2 diabetes management
• Chronic weight management in qualifying adults
Published trial data reported statistically significant outcomes in glucose markers and body weight reduction when used under medical supervision.
Because of these findings, semaglutide has become one of the most widely discussed GLP-1 receptor agonists in modern metabolic medicine.
This national attention naturally influences conversations in growing regions like St. George and greater Southern Utah, where healthcare infrastructure and wellness clinics have expanded in recent years.
FDA-approved formulations of semaglutide are indicated for:
• Type 2 diabetes management
• Chronic weight management in qualifying patients
These medications require prescription and physician oversight.
It is important to distinguish between:
• FDA-approved pharmaceutical products
• Compounded medications
• Research-grade materials
Each exists within different regulatory frameworks.
Beyond approved uses, GLP-1 receptor pathways remain an active area of metabolic research.
Researchers continue to study:
• Long-term metabolic signaling
• Cardiovascular outcome data
• Hormonal feedback loops
• Comparative incretin pathways
This broader scientific exploration contributes to continued academic and laboratory interest nationwide — including among research communities in Southern Utah.
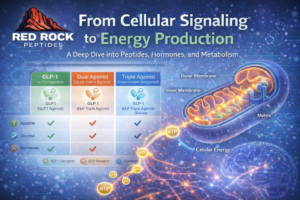
As research expands, semaglutide is often discussed alongside:
• Tirzepatide (dual GIP/GLP receptor activity)
• Retatrutide (triple-agonist under investigation)
Each compound interacts differently with incretin pathways. Comparative studies examine variations in receptor activity, pharmacodynamics, and clinical trial outcomes.
Understanding these mechanistic differences is essential for accurate scientific discussion.
In areas like St. George and Washington County, conversations around semaglutide often arise because:
• National media coverage has increased awareness
• Local healthcare providers discuss GLP-1 medications in appropriate clinical contexts
• Wellness trends spread rapidly in growing communities
However, public discussion does not replace medical evaluation.
Anyone considering prescription therapies should consult a licensed healthcare provider.
This distinction is critical.
Prescription semaglutide:
• Requires physician oversight
• Is regulated as a pharmaceutical product
• Is dispensed through licensed pharmacies
Research-grade semaglutide:
• Is intended strictly for laboratory research
• Is not approved for human consumption
• Must be handled in accordance with laboratory standards
Understanding this difference protects both consumers and suppliers.
Southern Utah’s population growth, active lifestyle culture, and expanding healthcare access contribute to heightened awareness of metabolic therapies.
As national research and clinical developments evolve, local curiosity often follows.
Education rooted in scientific accuracy is more valuable than hype.
In clinical settings, semaglutide is prescribed by licensed providers based on individual medical evaluation and FDA-approved guidelines. Accurate understanding of these regulatory frameworks helps ensure informed and responsible discussion.
Research-grade materials are intended for laboratory research purposes and are distinct from FDA-approved pharmaceutical products prescribed by licensed healthcare providers.
Large-scale clinical trials and expanded FDA approvals have increased national media attention.
Semaglutide represents one of the most studied GLP-1 receptor agonists in recent metabolic research.
For residents of St. George, Washington, Hurricane, Santa Clara, Ivins, and surrounding Washington County communities, understanding the scientific and regulatory distinctions is essential.
Accurate information — not marketing — should guide any conversation involving metabolic therapies.